In aseptic processes, being able to test the integrity of sterilising-grade filters, whether hydrophilic or hydrophobic, is critical. Indeed, even the slightest failure, if not detected, may compromise the sterility of the final pharmaceutical product, with regulatory and economic consequences as a result. How can filter integrity be tested? What equipment should be implemented?
Why test filter integrity?
Testing sterilising filters before use ensures their integrity prior to filtration of a batch, in order to identify any initially defective filters. Checking their condition after use ensures that their integrity has not been altered during filtration. In the latter case, detection of a defective filter will alert operators immediately after processing the pharmaceutical batch; they will then be able to carry out a new treatment after replacing the filter.
Pharmaceutical process engineers seek to implement non-destructive integrity tests for sterilising filters. If the tests must be carried out in-line, the procedure is even more demanding: the testing equipment must meet the aseptic conditions prevailing in the area and be easily movable from one filtration zone to another.
Sterilising filters are numerous on a pharmaceutical production line. These may include filters intended to ensure product sterility during production—whether for product components during addition, injected gases, or vents on tanks or containers used for storage, transport or final packaging. It is therefore also essential that the time required to perform a test be as short as possible, in order to limit the impact on production.
It should be noted that these physical integrity tests must be correlated with the bacterial challenge test for the filter, once confirmed intact, to be considered truly sterilising.
To find out more about sterilising filtration and bacterial challenge testing
How to test filter integrity?
There are three main families of non-destructive measurements used to test filter integrity:
- bubble point test (BPT, Bubble Point Test),
- diffusion test (DFT, Diffusion Flow Test)
- water intrusion test (WIT, Water Intrusion Test)
For hydrophilic filters, bubble point and diffusion tests are the most commonly used, with pressure hold and pressure decay tests as variants.
For hydrophobic membrane filters, the water intrusion test is generally used.
Bubble point test
The most frequently used non-destructive integrity test is the bubble point test. It is based on the fact that, in a filter, part of the liquid is retained within the pores by surface tension and capillary forces. When the filter is pressurised, a point is reached at which the pressure is sufficiently high to completely expel the trapped liquid and fully open the air pathways through the filter. A steady stream of bubbles is then observed at the outlet. The pressure at which this constant flow is observed is known as the bubble point. It depends on pore diameter. The integrity test thus makes it possible to detect minor defects in the filter and improperly sized pores.
In practice, to perform an integrity test, the filter is wetted with the appropriate test liquid (water for hydrophilic membranes or an alcohol/water mixture for hydrophobic membranes). The system is then pressurised up to 80% of the bubble point pressure value specified by the manufacturer. A bubble point value lower than the manufacturer’s specification will be observed if the filter is non-compliant or damaged.
Diffusion test
When the pressure is lower than the bubble point pressure, gas molecules migrate through the water-filled pores of a wetted membrane according to the law of diffusion (Fick’s law). This law states that, for a given filter, the diffusion flow rate is proportional to the applied pressure and to the total filter surface area.
To test filter integrity, the gas diffusion flow rate through the filter membrane is therefore measured at a pressure of approximately 80% of the expected bubble point. A diffusion flow rate higher than the specified limit indicates that the filter is damaged.
Pressure hold test
Also known as the pressure decay test, it has the advantage of presenting no risk to the downstream side of the installation. On the other hand, to measure pressure variations generated by gas diffusion through the media, it requires knowledge of the upstream volume in addition to the diffusion flow rate.
Water intrusion test
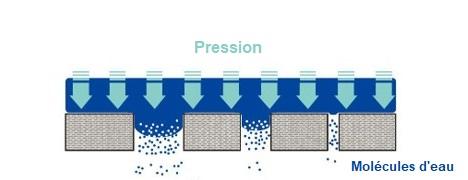
For this final test, the upstream side of the filter is brought into contact with ultra-pure water, before pressurising the filter with compressed air or nitrogen. It avoids the use of solvents such as isopropanol for hydrophobic sterilising filters. This test, which is particularly sensitive to operating conditions, measures the volumetric flow rate of water penetrating the hydrophobic membrane. The test pressure is selected so that water penetrates the surface of the filter membrane without fully wetting it. The pressure at which water passes through the pores of a hydrophobic filter is inversely proportional to pore size.
Portable testers: a turnkey solution
Integrity testing equipment must comply with 21 CFR Part 11 for tests performed in a pharmaceutical environment. Portable and compact devices are available, allowing bubble point tests, diffusion flow tests and water intrusion tests to be carried out in the laboratory or in situ.
The lightest units may weigh less than 10 kg.
They allow testing of both hydrophilic and hydrophobic membrane filters, as well as filter cartridges and capsules. Mass flow measurement makes it possible to eliminate the influence of temperature and pressure variations.
Easy to use, they can be deployed in sterile areas (stainless steel housing).

Contact a filtration expert
We are at your service
Our experts place their knowledge and expertise at your disposal for liquid filtration, air and gas filtration, decontamination, and separation technologies.

