Since the Second World War, industry has been producing filtration membranes capable of retaining, through filtration, particles as well as free micro-organisms in a liquid.
“Sterilising” membrane: definition
These so-called “sterilising” filtration membranes (0.2 µm or 0.22 µm filtration rating) can therefore act as a barrier to micro-organisms by preventing them, without destroying them, from progressing further through the system. The micro-organisms thus accumulate on the filtration membrane. This widely used technology is highly effective, but time-limited, as the micro-organisms remain alive on the medium.
After a few hours, or at most a few days, the phenomena known as “go-through” (pass-through), “grow-through” (growth through), and “blow-through” (penetration through) begin to appear. The thicker the filtration membrane, the more stress phenomena are generated at the bacterial level for an equivalent porosity, thereby amplifying the “go-through” and “blow-through” effects.

The FDA (Food and Drug Administration) defines these filters as “a filter which, when tested with a suspension of Pseudomonas (Brevundimonas) diminuta containing a minimum of 10⁷ organisms per cm² of filter surface, produces a sterile effluent.”
These filtration membranes are generally validated using the HIMA challenge. The exact procedure for this official test was defined by the Health Industry Manufacturers Association (HIMA) and is described in “Microbiological Evaluation of Filters for Sterilizing Liquids” (HIMA document #3, Vol. 4, 1982). Since the 1960s, occasional contamination issues—other than those caused by mycoplasma—have been reported despite the use of 0.2 µm and 0.22 µm “sterilising” membrane filters.

Consequently, the FDA recognised that 0.2 µm and 0.22 µm membrane filters cannot effectively prevent filtrate contamination. In 1996, it acknowledged that the use of 0.1 µm filters as final filters is “preferable to ensure product sterility…”.
However, the risk then is to limit flow rate, as it is generally considered that there is an average 40% loss of flow when replacing 0.2 µm sterilising filters with 0.1 µm filters of equivalent size and filtration surface area.
In addition, some flat membrane manufacturers use the “subterfuge” of a positively charged membrane surface to achieve bacterial retention sufficient to meet the HIMA challenge, without penalising flow rate and therefore by using larger porosities. During tests with latex microspheres (particulate testing), electron microscopy reveals that adhered microspheres are then found on both sides of the membrane.

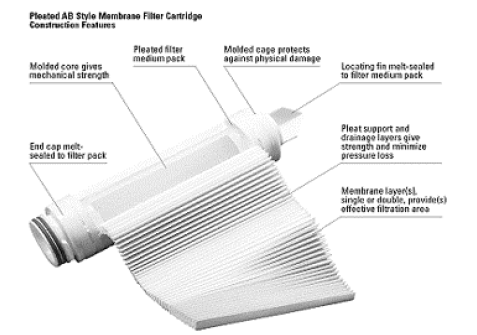
Flat membrane technology
These filtration membranes have existed for more than 70 years, making them a well-known and proven technology. To achieve an acceptable flow rate, the filtration surface area must be increased as much as possible.
The membrane is therefore pleated before being inserted into the cartridge, making it possible to obtain surface areas between 0.5 m² and 0.7 m². This surface area naturally remains the limiting factor for flow rate. The most commonly used media are polyethersulfone, nylon 6.6, or PVDF. The thickness of these membranes ranges between 150 and 300 µm.
To improve the mechanical strength of these filtration membranes, they are generally sandwiched between two support and drainage layers, usually made of polypropylene. The centre of the cartridge consists of a rigid perforated core, combined with an external cage, providing the cartridge with good mechanical strength and high pressure resistance.
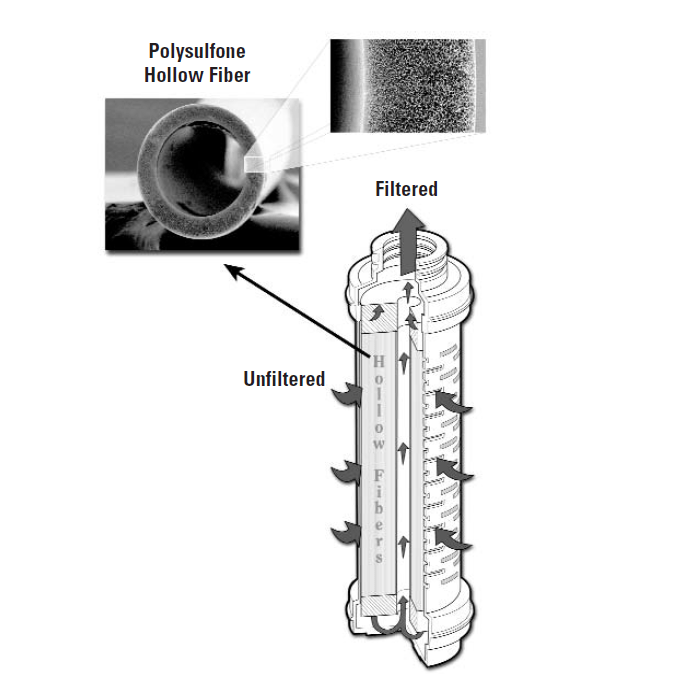
Hollow fibre technology

Rather than using pleated membranes, which are inherently limited in filtration surface area, these filters employ a hollow fibre membrane (FIBERFLO).
These membranes are widely used in dialysis filters, and increasingly in the field of separation technologies (microfiltration or tangential ultrafiltration).
Depending on the manufacturer, these fibres have different diameters, as well as different media qualities (polysulfone, nylon, PVDF, polypropylene).
This technology offers the major advantage of multiplying the filtration surface area by two or even three for the same filter element size, compared with a pleated membrane cartridge. A 10-inch filter element, for example, provides a filtration surface area of 1.5 m².
This larger filtration surface area then makes it possible to reduce the filtration rating without loss of flow rate, thereby increasing filtration efficiency (for example, using 0.1 µm instead of 0.2 µm), or to increase flow rate without affecting pressure drop.
While remaining compliant with the same standards, a 0.1 µm filter will therefore deliver the same flow rate as most 0.2 µm filters of equivalent size.
Why is flow rate important?
Any process, whatever its nature, requires a reliable and defined feed flow rate. In this respect, filter sizing can be critical. If it is undersized, the filtration stage will generate significant pressure drops, which will increase exponentially as the filter cartridge becomes fouled.

A tighter sterilising filtration therefore provides a higher level of assurance for the protection of a purified water production and distribution system.
One of the specific features of FIBERFLO filters is that they are initially validated using a particulate challenge, and subsequently validated according to the HIMA challenge.
The particulate challenge is absolutely non-permissive, as all calibrated microspheres must be retained by the filter.
Consequently, the declared retention rating is truly absolute. The HIMA challenge, for its part, only requires a 7 log/cm² reduction on the reference micro-organism.
In practice, FIBERFLO hollow fibre filters make it possible to:
- Increase flow rate for the same filtration rating without compromising retention and without having to add or replace the filter housing
- Improve bacterial and particulate retention without compromising flow rate
A particular application of FIBERFLO hollow fibre filters is endotoxin retention in the pharmaceutical field.
Learn more about how to remove endotoxins from pharmaceutical-grade water


Contact a filtration expert
We are at your service
Our experts place their knowledge and expertise at your disposal for liquid filtration, air and gas filtration, decontamination, and separation technologies.