Industry sector: Pharma / Healthcare
Company activity: Manufacturer of dermatological products
Company type: Major European manufacturer of pharmaceutical and dermo-cosmetic products
The filtration of parenteral medicinal products requires strict validation protocols to ensure product safety. Likewise, topical treatments, which are non-prescription, for alopecia or male pattern baldness, require equally appropriate levels of safety.
Filtration challenge
A major manufacturer of an alopecia treatment based on the API Minoxidil uses the SupaGard filter from AMAZON FILTERS to clarify the final product. While the quality of the SupaGard filter is supported by a comprehensive validation guide and is manufactured from materials compliant with FDA CFR 21 and USP Class VI Plastics, a recent audit by regulatory authorities highlighted the need to carry out additional testing.
Validation guide
SupaGard cartridges are used in highly specific applications and must therefore comply with strictly defined manufacturing and quality standards. The Validation Guide describes the characteristics of the filter cartridge and the manufacturing and quality standards, in particular to demonstrate product compliance with the requirements of regulatory authorities.
The auditors required documentary evidence certifying that the filter could not alter both the quality and the safety of the finished product, in compliance with the specific parameters of the process.
Pemflow filtration solution
Our approach

Often, data relating to extractables and the protocols to be implemented are not clearly defined by regulatory bodies. As a result, the customer needs to be supported in conducting validation testing and in providing the full set of documents describing the protocols and test results.
To achieve this, we work with a network of experienced laboratories, specialized in the validation of cGMP filters (current Good Manufacturing Practice), in close collaboration with the customer. This partnership enables us to establish relevant protocols and draft final validation reports that can be submitted directly to regulatory authorities. This approach gives us access to a substantial knowledge base for the development of protocols with short turnaround times.
Protocol development
The objective was to demonstrate that no contamination was extracted from the cartridge during product filtration. To this end, a solvent was developed, based on the product formulation, to replicate the behavior of the API-containing solution during filtration. To simulate the worst-case conditions under which validation was performed, the solution was heated to a temperature 5°C higher than that of the production process.
The specific solvent, at a 5% API concentration, is illustrated in the table below:
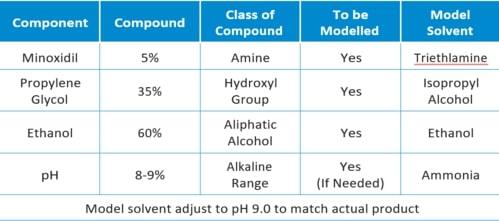
Results
Three tests were performed. The specific solvent was filtered under conditions identical to those of normal production.
Non-volatile residues (NVR) were quantified, as well as extractables identified by chemical analysis (FTIR technique: Fourier Transform Infrared Spectroscopy).
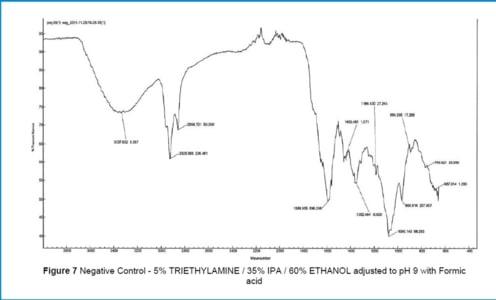
Conclusion
It was demonstrated that the compounds extracted from the SupaGard cartridge from AMAZON FILTERS, even under worst-case conditions, consist solely of filter construction materials (polypropylene) and at extremely low levels (10 µg/L of filtered product).
The filter therefore does not affect the quality of the specific product. The fact that no foreign contamination was detectable on the cartridge demonstrates the overall cleanliness levels of the filter elements supplied to the customer.
The final report was readily accepted by regulatory authorities as evidence of the compliance of SupaGard filter cartridges with respect to the process and its constraints.
We are able to take full responsibility for the validation of our filter elements for specific products and processes, and to provide the documentation required by regulatory authorities as proof of compliance with your requirements and expectations.

Contact a filtration expert
We are at your service
Our experts place their knowledge and expertise at your disposal for liquid filtration, air and gas filtration, decontamination, and separation technologies.

