Industry sector: pharmaceutical laboratory
Company activity: production of medical-surgical equipment for the treatment of chronic and acute renal failure
Company type: major global group
Company founding period: 1930s
Contacts: Buyer EMEA – Process Manager: ETO (Ethylene Oxide) Sterilization, AN69 Polymer & Membrane Workshop
Company context
The company designs, develops, and produces medical-surgical equipment (hemodialyzers and sterile single-use hemofilters) for the treatment of chronic and acute renal failure.
It uses filters on its production lines to produce pharmaceutical-grade water.
Discover two articles you may find useful on pharmaceutical waters:
Regulatory constraints on the quality of pharmaceutical-grade water
Decontamination of pharmaceutical-grade water
Filtration challenge
The filtration systems had to limit the presence of endotoxins in the demineralized water. These molecules, located in the outer membrane of certain bacteria (E. coli, Salmonella), are released when the bacteria are destroyed. They are recognized and attacked by the human immune system. Exposure to these substances can have severe, even lethal, consequences.
The company sought to implement a filtration solution capable of reducing endotoxin concentration from 10 to 1 EU/mL in demineralized water at 15 °C under 3 bar pressure, before it entered the tank. The required treated flow rate was 5 m³/h, for approximately 20 hours per day.
To learn more about endotoxin filtration, see our article How to remove endotoxins from pharmaceutical-grade water?
Pemflow filtration solution
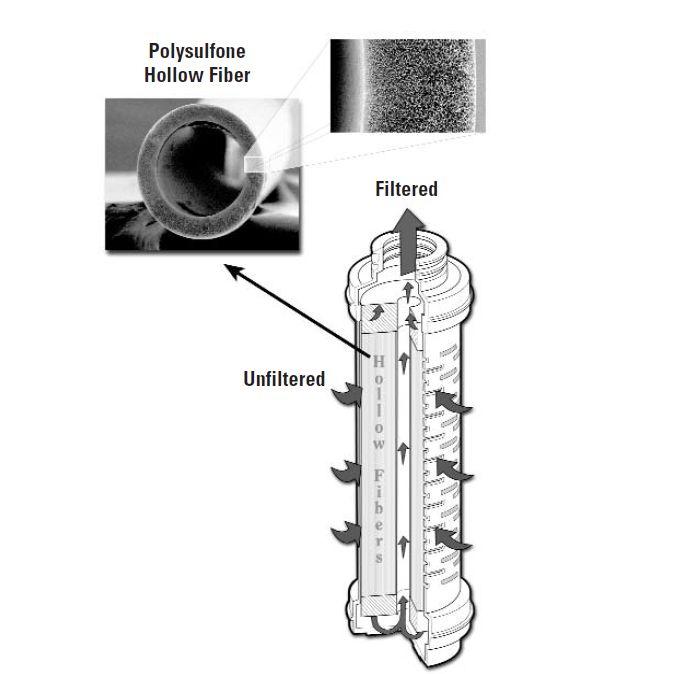
We recommended the use of hollow-fiber filters, the most suitable technology for endotoxin retention.
To meet the performance targets defined in the customer’s specification, we installed a housing containing five 30″ filter cartridges. We also selected a flow rate that would ensure sufficient contact time between the water and the filter media, namely 8 L/min per 10″ height.
The characteristics of the selected filter cartridges are as follows:
- filter media: polysulfone hollow fibers
- core, cage, and end caps: polypropylene
- fiber potting material: polyurethane
- length: 30″
- end caps: code 7
- absolute filtration rating: 0.05 μm
- filtration surface per cartridge: 4.5 m²
- steam-sterilizable
- endotoxin retention: 5 EU/mL

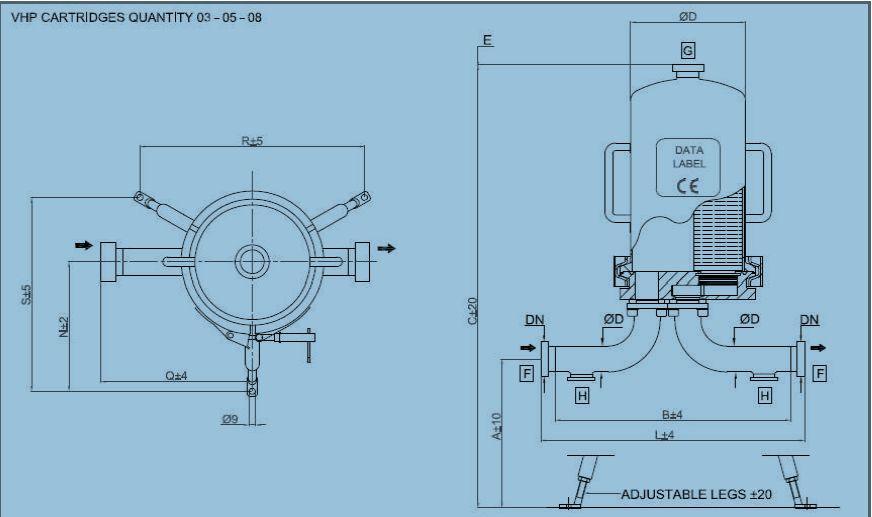
The cartridge housing has the following characteristics:
- housing: 316L stainless steel – for 5 × 30″ cartridges – Code 7
- number of cartridges: 5
- operating temperature range: –20 / +150 °C
- operating pressure range: –1 / +10 bar
- inlet/outlet: 2″
- Tri-Clamp purge: no vent: 1.5″
- Tri-Clamp finish: mechanical polish Ra < 0.8 μ
- ATEX: Group 2, Category 2 Gas & Dust, Fluid GR IIC, Zone 1/2
- PED: Category 2
This solution successfully reduced the endotoxin level below the specified limit.

Contact a filtration expert
We are at your service
Our experts place their knowledge and expertise at your disposal for liquid filtration, air and gas filtration, decontamination, and separation technologies.