Over recent years, the European Union has progressively tightened restrictions on per- and polyfluoroalkyl substances (PFAS) to limit their impact on the environment and human health.
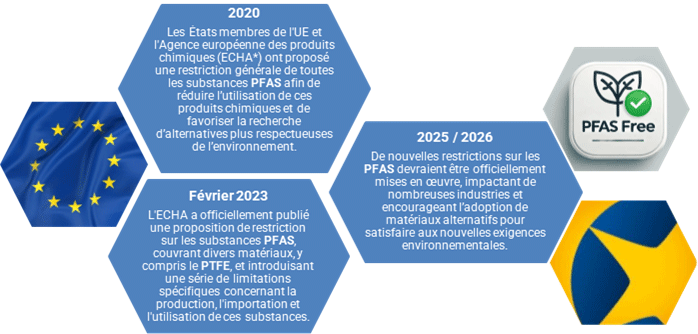
PFAS are widely used across many industrial sectors and consumer applications for their non-stick, waterproofing and high-temperature resistance properties. However, these chemicals, which are resistant to degradation, lead to persistent pollution and tend to bioaccumulate, prompting increased regulatory scrutiny.
PTFE (Polytetrafluoroethylene) and PVDF (Polyvinylidene fluoride), widely used in filtration, are also types of PFAS. In order to comply with regulations and reduce environmental risks, the adoption of PFAS-free materials as alternative solutions has become a dominant trend.
PES (Polyethersulfone), as an alternative solution, offers remarkable chemical compatibility and excellent mechanical performance, while being PFAS-free; making it a preferred choice for sustainable development, while complying with today’s and tomorrow’s regulations.
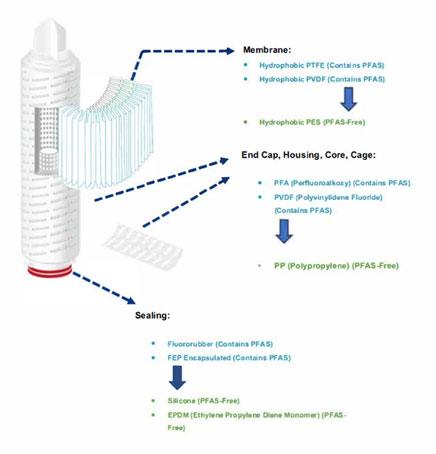
Fields of application
1. Sterilising filtration applications for compressed air and oxygen
2. Sterile gas inlets and outlets for fermentation vessels, liquid storage tanks and mixing vessels
3. Sterilising filtration applications in single-use systems
4. Sterilising filtration applications for sterile filling gases
PES vs. PTFE/PVDF
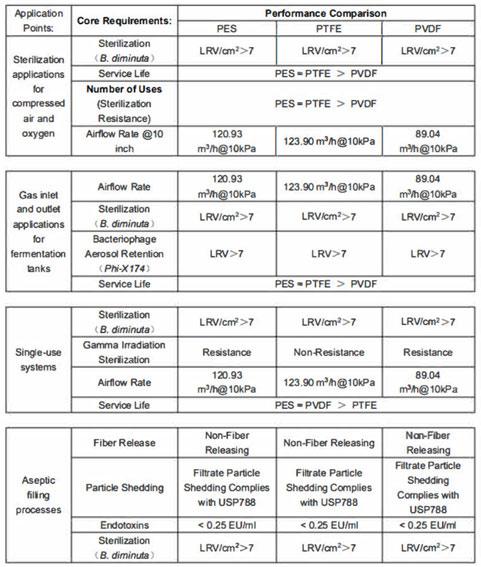
This table shows that Polyethersulfone (PES) cartridges deliver airflow performance comparable to that of Polytetrafluoroethylene (PTFE) and Polyvinylidene fluoride (PVDF) cartridges. In addition to their excellent steam resistance, these hydrophobic PES filters demonstrate gamma irradiation resistance equivalent to that of hydrophobic PVDF filters, making them particularly well suited for applications in single-use systems.
Regulatory compliance
It is recommended that these PES membrane cartridges meet several standards and requirements:
- Complies with the “non-fibre releasing” filter definition specified in 21 CFR 210.3 (b) (6).
- Insoluble particles in the filtrate comply with USP <788>.
- Bacterial endotoxins comply with USP <85>.
- Bacterial retention validation complies with ASTM F838 standards.
- Retention efficiency for bacteriophage Phi-X174 (ATCC ® 13706-B1) exceeds 99.99999%.
- All component materials comply with FDA requirements for indirect food additives as defined in 21 CFR Parts 177–182.
- Biocompatibility complies with USP <87> and USP <88>.
- All components of this product are manufactured from materials of non-animal origin.

Contact a filtration expert
We are at your service
Our experts place their knowledge and expertise at your disposal for liquid filtration, air and gas filtration, decontamination, and separation technologies.

